
Department of Toxicology and Cancer Biology,
College of Medicine, University of Kentucky
University of Kentucky Markey Cancer Center
Health Sciences Research Building 461
1095 Veterans Drive
Lexington, KY 40536-0305
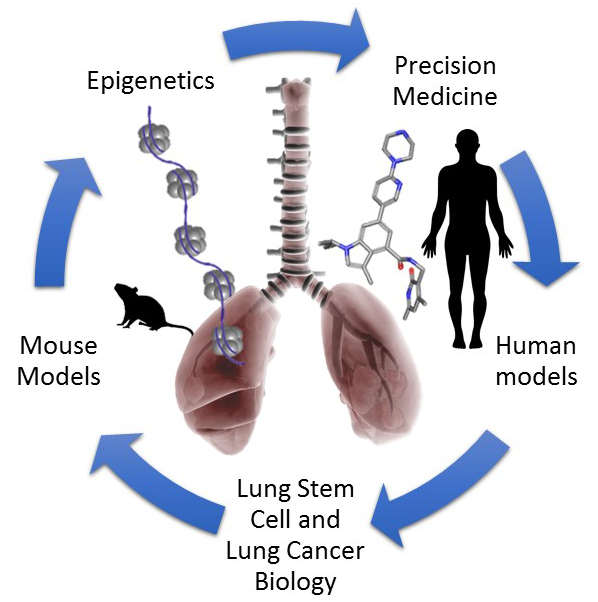
The overall goal of the Brainson Lab is to define precision therapy options for genotype- and subtype-specific lung cancer using ideas and techniques from stem cell biology and epigenetic research.
Non-small cell lung cancer (NSCLC) is a devastating disease with extensive genetic, cellular and epigenetic complexity. For example, an average human lung tumor has over 200 non-synonymous mutations and can contain cells that have markers of several different lung lineages. The lineage relationship and epigenetic differences between the two major subtypes of non-small cell lung cancer, namely lung adenocarcinoma (ADC) and lung squamous cell carcinoma (SCC), are unclear.
The mammalian lung is a complex organ containing numerous stem/progenitor cell populations, any of which may serve as cells-of-origin for lung carcinomas in various genetic and microenvironmental contexts. Our lab is directly testing cell-of-origin fitness by isolating lung stem and progenitor cells and introducing specific genetic events into the isolated populations. During the transformation process, we culture the cells in 3-dimensional organoid culture systems. Once genetically modulated organoids are established, they are then implanted into immunocompomised mice to assess malignancy and tumor subtype. Using this system, the epigenetic changes that cells undergo during transformation will be assessed. Understanding which epigenetic states are permissive for lung cancer initiation, and in which microenvironmental contexts, will help us to target the root causes of the disease. In addition, studying the earliest stages of disease may suggest new methods of early detection for lung cancer.
While our cell-or-origin approaches will give valuable insight into the early stages of lung tumorigenesis, more clinically important is finding methods to incorporate epigenetic inhibitors to treat established lung tumors. Therefore, our lab uses primary tumors from genetically engineered mouse models (GEMMs) of lung cancer to explore the effects of epigenetic inhibitors on tumor cell growth, differentiation and response to other chemotherapies. While many lung cancer cells cannot grow as 2-dimensional cultures, we can grow cells of diverse genotypes, including KRASG12D, KRASG12D/Lkb1-null, EGFRT790M/L858R and Lkb1-null/Pten-null in 3-dimensional cultures. Using these cultures, our main focus is to link the genotype and subtype of the lung tumor cells to efficacy of the epigenetic inhibitors. To validate our findings in human tumors, we are also establishing several patient-derived organoid models of lung cancer.
Lung adenocarcinomas tend to arise in more distal alveolar space and have glandular features and markers consistent with distal lung, including surfactant protein C (SPC) and the transcription factor TTF1. Lung squamous cell carcinomas arise in the more proximal airway and are characterized by fully stratified squamous epithelial layers of cells. Many questions exist about these two types of non-small cell lung cancer, and our laboratory aims to address these questions.
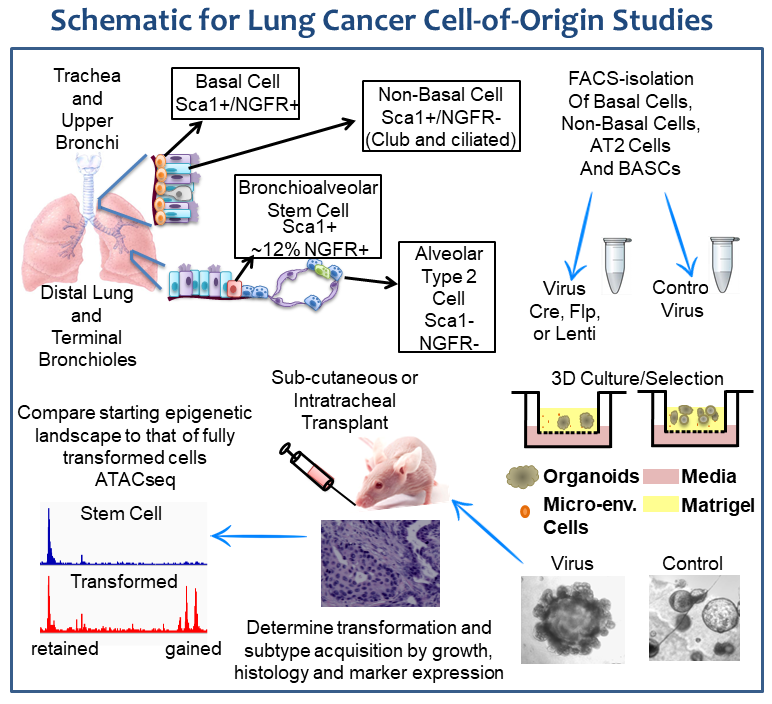
After isolation by fluorescence activated cell sorting (FACS), the genetics of the normal lung cells are modulated and the cells are grown in 3-dimensional organoid culture systems. In these cultures, cells grow in Matrigel that is exposed to air from above and liquid media below. This air-liquid-interface type culture, along with the specialized media and support cells that we use in the cultures, allow the cells to retain lung specification. We have successfully transformed several lung cell types, as judged by growth of tumors after cell implantation into immunocompromised mouse lungs. Our current studies are interrogating the epigenetic changes that take place during the transformation process. Wiggle plots and micrographs are for illustrations purposes only.
Primary tumor cells of known genotypes are isolated from GEMMs and cultured in 3-dimensional culture systems. Epigenetic inhibitors are applied to cultures during serially passaging. Therapies that work well to inhibit tumor organoid growth will be further explored for mechanism of efficacy and in vivo efficacy. Our goal is to identify epigenetic therapies that target specific genotypes and subtypes on non-small cell lung cancer, with the end goal of finding new precision medicine options for patients. Wiggle plots and micrographs are for illustrations purposes only.
The overall goal of the Brainson Lab is to define precision therapy options for genotype- and subtype-specific lung cancer using ideas and techniques from stem cell biology and epigenetic research.

cfbrainson@uky.edu
Born and raised in Massachusetts, Chris has always wanted a career in cancer research. Chris joined the faculty of UK in the Toxicology and Cancer Biology department in October 2016. She previously worked as a post-doctoral fellow at Boston Children's Hospital, where she was part of the Harvard Medical School Department of Genetics, the Boston Children's Hospital Stem Cell Program, and the Harvard Stem Cell Institute. Her research focuses on defining precision medicine opportunities for lung cancer by leveraging ideas and techniques from stem cell biology and epigenetics. Her research is currently funded through the NCI K22 career transition award, and the American Cancer Society.
plaugher_dr@uky.edu
Daniel is from rural West Virginia (the state, not the western territory of Virginia), where he grew up on his family's cattle farm that also raises goats, pigs, chickens, horses, rabbits and more. He was a multi-sport, all-conference, all-state, and multi-time state-champion track athlete, eventually becoming a nationally competitive collegiate high hurdler at West Virginia Wesleyan College. He always had a love for mathematics and teaching, which lead him to earn B.S./B.A degrees in mathematics and mathematics education (5-Adult) as a first-generation college student. Daniel earned his M.S/Ph.D. degrees at the University of Kentucky, specializing in mathematical oncology. While doing so, he helped construct a strategic pipeline utilizing tools used for developing and analyzing dynamical models. In particular, he specializes in building stochastic Boolean networks that can be used for creating computational models of patient-specific cancers. To find out more, you can visit his personal website at https://sites.google.com/view/daniel-plaugher.

xso224@uky.edu
Xiulong grew up in China’s countryside where he first became interested in studying biology, and earned his degree in Veterinary Medicine. He then decided to move to the United States to research, study, and explore the mechanisms behind diseases and their treatments. Xiulong attended the University of Rhode Island where he obtained his Ph.D. in Pharmacology and Toxicology. In order to further pursue his research interests of virology, immunology, drug metabolism and toxicity, endocrinology, and cancer biology, he accepted a position as a post-doctoral scholar in the Brainson lab, where he is exploring the epigenetic mechanisms behind lung cancer initiation and development. His interests outside of the lab include playing ping pong and badminton.
averychill20@uky.edu
Avery was born in Louisville, Kentucky. After moving around quite a bit he finally ended up back in the bluegrass at University of Kentucky doing his undergraduate studies in Kinesiology and minoring Biology. He then completed his Masters in Biomedical Sciences where his passion for science was cultured. Avery was then introduced to the different options available at the University of Kentucky and became a PhD student in 2022 and officially joined the lab in the same year. Outside of the lab, he enjoys snowboarding, working out and hiking.
davepreston.esoe@uky.edu
Dave was born and raised in Atlanta, GA till he turned 10 and moved to Cameroon where he lived for 8 years. He came back to the USA for college and attended Bemidji State University where he obtained a B.S. in Biology in the spring of 2020. That fall (2020), he enrolled in a Masters program at the University of Kentucky studying Forensic Toxicology and Analytical Genetics with emphasis in Analytical Genetics. During his time as a Masters student, he developed an interest in studying epigenetic regulation and disease development, so decided to apply to the department's PhD program. After his PhD, he would love to work in the genomics field, specifically in a clinical setting investigating genetic and epigenetic causes of life-threatening diseases.
christiangosser@uky.edu
Christian is a native Hoosier from Rushville, Indiana. Throughout his entire life he liked science and originally wanted to study space and astronomy but eventually found his liking for biology after AP Biology in high school. For his undergraduate degree, he went to Wabash College and got his Bachelor of Arts in Biology in 2020 where he focused on Genetics and Ecology, but ultimately chose to pursue genetics as his future career. He then found a Masters program centered around Forensic Science and Genetics where he earned his MSc in 2022 and would learn about cancer biology and come to learn about pharmacology and genetics together. His future career and research interests are based around pharmacology and precision medicine treatment in cancer and to help research more efficient treatment options. Christian in his free time is an avid golfer and has been playing since he was 12 and has been playing saxophone since he was 10.
yindan.lin@uky.edu
Yindan was born and raised in Guangzhou, China, and earned her bachelor’s degree from Guangdong Pharmaceutical University in 2018. She then worked as a research assistant at Zhejiang University and the Icahn School of Medicine at Mount Sinai, where she focused on cancer research and antibody engineering for targeted therapies. Motivated by a strong aspiration to pursue advanced education, Yindan began her Ph.D. studies at the University of Kentucky in the fall of 2024 and officially joined the Brainson Lab in 2025. Her research investigates how epigenetic inhibitors can be leveraged to treat pulmonary fibrosis under hypoxic conditions. Outside the lab, Yindan enjoys hiking, reading psychology, and traveling.
yzhao@uky.edu
Yanming has extensive experience in cancer biology research and has worked in the UK system for over 14 years. She currently splits her time between the Brainson lab and the Rangnekar lab, and has also worked and published with the St. Clair lab.
abigail.edgin@uky.edu
Abby graduated in May 2017 from the University of Kentucky with a Bachelor’s of Science degree in Biology and a minor in Italian Studies. She is currently in Medical School at the University of Louisville. When she isn’t in the lab, Abby enjoys running and cheering on the Cats at sporting events. After the lab, Abby went to University of Louisville medical school after graduation.
alu234@uky.edu
Born in Belarus and immigrated to the US as a child, Alex has always loved science and medicine. Currently working on his B.S. in Chemistry here at the University of Kentucky, Alex has joined the Brainson lab to learn more about cancer biology, see how the world of academic research works, and help contribute to the field of medicine in any way he can. He plans on continuing his education after his bachelor’s degree by applying to medical school. When he is not in class or in the lab, Alex enjoys cooking and painting to unwind. After the lab, Alex went to University of Kentucky medical school after graduation.
john.fan.chen@uky.edu
Fan is a graduate student who joined the lab in March of 2017. Previously, he received his M.D. degree and medical license from Shandong University, China. Aiming to be an oncologist in the future, he is focusing on the epigenetic precision medicine for lung cancer, specifically on Ezh2and PRC2 function. After the lab, Fan went to Sun-Yat Sen Cancer Center to do his internship and residency.
aria.byrd@uky.edu
Aria Byrd is an Atlanta, Georgia native who received her B.S. in Biology with a concentration in biotechnology from Albany State University, and her M.S. in biology from North Carolina Agricultural and Technical State University. Her growing fascination with understanding how the body responds to both voluntary and involuntary environmental exposures has led her to pursue a doctoral degree in Toxicology. In the Brainson lab, she is acquiring advanced laboratory and research training that will prepare her to effectively investigate environmental perturbations that affect human health. Her long-term goal is to conduct research on underserved populations, specifically, to investigate the physiological changes that are currently taking and have taken place as a result of environmental stressors. Her life-long aspiration is to play an integral role in influencing national and global health policy that better cater to underserved populations. After the lab, Aria went to Emory University to be a project manager.
tannerjducote@uky.edu
Tanner joined the lab in June of 2018 after completing his first-year rotations in the IBS program at the University of Kentucky. Tanner is a native of Lafayette, Louisiana (the Heart of Cajun Country) and completed his bachelor’s degree in Biology, focused in Microbiology, from the University of Louisiana at Lafayette. During his undergraduate studies, he developed a passion for understanding the fundamentals of the immune system and the therapeutic implications in the context of cancer. While in the lab, Tanner is focused on understanding how the immune microenvironment, within tumors of the squamous cell carcinoma phenotype, play a role in the progression and treatment of the disease. More specifically, he is currently working toward understanding the biology of tumor associated neutrophils (TANs) and their role in suppressing the activation and function of CD8+ T-cells. Utilizing the information gained by studying these TANs, Tanner hopes to increase the efficacy of current immunotherapies, such as the anti-PD1 immune checkpoint inhibitor and the underlying mechanisms of synergy with the inhibition EZH2. When not in the lab, Tanner is actively involved in community outreach and also enjoys cooking, symphonic performances, and hiking the local mountain trails. After the lab, Tanner became a Molecular Science Liaison at Caris Life Sciences after graduating.
Hannah.Bowman@uky.edu
Hannah was born and raised in Lexington, KY. After high school, she decided to pursue a nursing degree because of her interest in science and patient care. Upon taking prerequisite anatomy & physiology for nursing school, she found an insatiable curiosity to learn the complex science behind how the human body operates. While pursuing her nursing degree at the University of Kentucky, she found her passion for health equity and clinical care but still held onto her affinity for the natural sciences by taking courses outside of the nursing school curriculum. In doing so, she took a biochemistry course, which introduced her to epigenetics. She was amazed by this topic and intrigued by the integral relationship it has with social determinants of health. Epigenetics was the piece of the puzzle that connected her love for science and her passion for addressing health equity for vulnerable populations which is what led her to Dr. Brainson’s lab. Hannah graduated with her BSN in 2021 and plans to apply to medical school in 2022. After the lab, Hannah became a medical student at Dartmouth College after graduating.
kass.naughton@uky.edu
Kassie grew up in Loveland, Ohio, just outside of Cincinnati. In high school, thanks to a class called Biotechnology, she learned she had a passion for research and science. Because of this experience, she attended The Ohio State University, where she studied chemistry. While at Ohio State, Kassie worked as an undergraduate researcher studying how the production of hydrogen gas from metalloproteins can be used as a fuel source. Due to her project involving nickel toxicity, Kassie became interested in pursuing a PhD in a medicinal field. She became a graduate student at the University of Kentucky in 2019 in Toxicology and Cancer Biology and began working on discovering the role of the G3BP1 protein in lung cancer progression. Kassie officially joined the lab in 2021 and is currently continuing to work with her project on the G3BP1 protein but is also finding ways in which it may be related to ongoing projects in the Brainson lab. Outside of the lab, Kassie enjoys traveling, reading, and her cat Betsy. After the lab, Kassie started working at MedPace Inc after graduating.
emskag2@g.uky.edu
Erika Skaggs is a non-traditional student with a baccalaureate in political science from the University of Kentucky and a master’s degree in security and intelligence studies from the Patterson School of Diplomacy and International commerce. She is currently studying chemistry and health, society and populations as an undergraduate and plans to attend medical school following graduation. As a physician and scientist, she would like to continue exploring the relationship between intelligence studies and clinical practice. Over the last several decades the United States’ defense apparatus has spent billions of dollars researching new instruments to confront an ever changing security landscape. Some of those technologies are ripe for exploitation in the iatric theater. In fact, researchers at the University of Exeter are already using AI to increase early detection of dementia, and even spot mistaken diagnoses. Erika would like to continue that trend by utilizing big data technologies to identify hidden trends in oncologic populations, and establish new targets for treatment and prevention. After the lab, Erika became a medical student at University of Cincinnati after graduating.Our lab is directly testing cell-of-origin fitness by isolating lung stem and progenitor cells and introducing specific genetic events into the isolated populations.
Understanding which epigenetic states are permissive for lung cancer initiation, and in which microenvironmental contexts, will help us to target the root causes of the disease.
Our goal is to identify epigenetic therapies that target specific genotypes and subtypes on non-small cell lung cancer, with the end goal of finding new precision medicine options for patients.
We are grateful to our sponsors







Studying the earliest stages of disease may suggest new methods of early detection for lung cancer.
We are always looking for motivated individuals to join our team. Please contact us here:
Fillmore Brainson Lab
Department of Toxicology and Cancer Biology, College of Medicine, University of Kentucky
University of Kentucky Markey Cancer Center
Lab Address:
Health Sciences Research Building 461
1095 Veterans Drive
Lexington, KY 40536-0305
Phone: (859)323-4973
Please use the form below to send an email to us. Please be sure to check the recaptcha box.